Malaria is a life-threatening disease caused by Plasmodium parasites that are transmitted to people through the bites of infected female Anopheles mosquitoes.
While the species Plasmodium falciparum causes 99,7% of the cases in the African region, Plasmodium vivax accounts for 74,1% of estimated malaria cases in the Region of Americas. Nevertheless, malaria disease is less virulent and mortal when caused by P. vivax than when caused by P. falciparum. For this reason, even if there are around 14 different Plasmodium species causing disease in humans, malaria is frequently just linked to Plasmodium falciparum1.
But how is malaria transmitted through a mosquito if it is in fact Plasmodium the parasite causing it?
Parasites are organisms that live inside or on the surface of a host organism, which they use to help them survive.
Plasmodium falciparum infects female Anopheles mosquitoes with gametocytes (the precursors of female and male gametes or sexual cells) when one of them bites an individual suffering from malaria. Indeed, mosquitoes and humans are the two Plasmodium hosts.
Once at the mosquito’s intestine, P. falciparum undergoes multiplication (in a cycle known as the sporogonic cycle). As a result of this multiplication, structures known as sporozoites are formed and released. When biting an individual, the mosquito injects the sporozoites to the human bloodstream.
At that point, the human becomes its new host and two cycles take place: the first one in the liver (known as the exo-erythrocytic cycle), and the second one on the bloodstream (known as erythrocytic cycle).
In the liver, sporozoites infect liver cells, mature and form schizonts. Once schizonts burst, they release merozoites.
In the bloodstream, P. falciparum’s merozoites infect host red blood cells as a strategy to avoid being recognised by macrophages that would kill them2. They also digest host erythrocytes’ haemoglobin, as they need it to grow and to complete asexual replication. Under the microscope, haemoglobin loss in infected red blood cells is translated in a loss of brightness and density, compared to healthy erythrocytes3 (Figure 1).
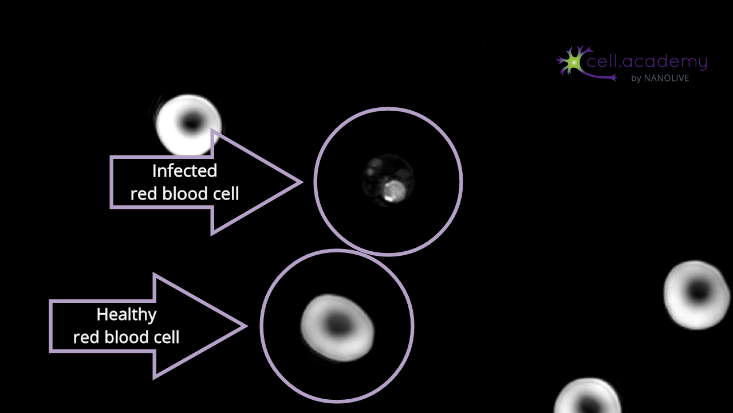
Figure 1. Caption of live cell video showing malaria blood sample (infected red blood cell surrounded by healthy red blood cells)
During the erythrocytic cycle, merozoites mature into rings and from this stage and on, there are two possible options: either they evolve to form gametocytes, or they evolve and form new merozoites through intermediate stages (trophozoites and schizonts). Gametocytes will be again ingested by Anopheles mosquitoes, leading to a continuation of P. falciparum’s life cycle4,5.
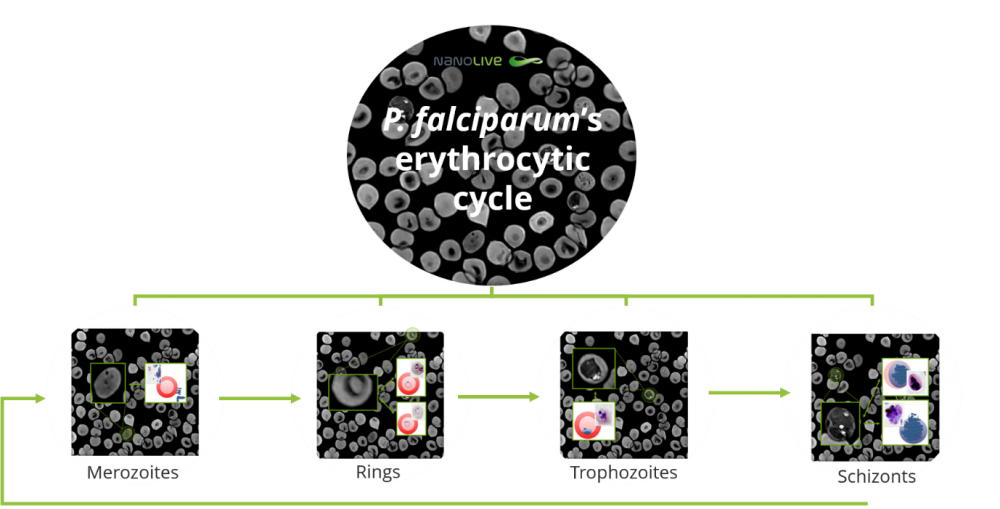
Figure 2. Plasmodium falciparum’s erythrocytic cycle seen under the microscope. The different stages are highlighted and compared with traditional microscopy examples.
To know more about malaria’s erythrocytic cycle and how to diagnose malaria under the microscope click here! And if you want to refresh your knowledge about the immune system in general and macrophages in particular, do not hesitate to read our post and watch the video!
What’s the situation today?
At present, malaria causes millions of cases per year. 219 million cases were registered in 2017, which lead to 435 000 deaths, 266 000 of which were children under 5 years.
92% of the total cases were registered in the African region, which also accounted for 93% of the total deaths. Doubtlessly, although there are 87 countries around the world that have registered malaria, the African continent is the one suffering the strongest disease burden1.
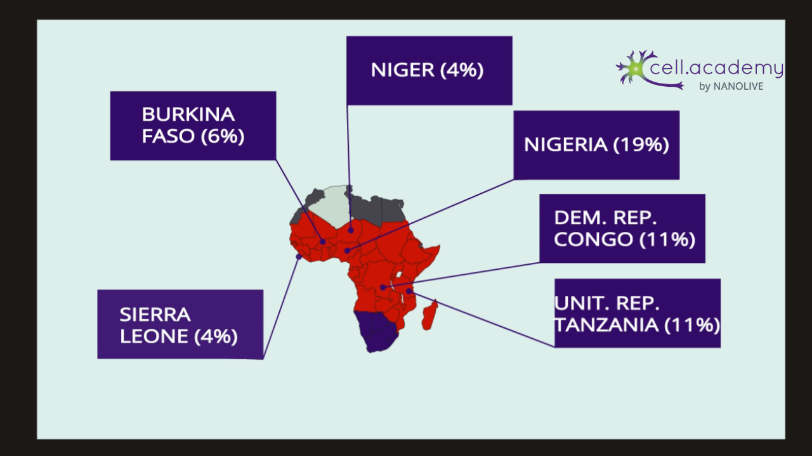
Figure 3. 93% of total malaria cases are in Africa, where only 6 countries account for 55% of the total malaria cases registered in 2017.
What is the plan for the future?
The World Health Organization, whose main concern is international public health, developed a Global Technical Strategy for malaria, which aims to reduce the number of cases and deaths related to the illness by 90%, and increase malaria-free countries with at least 35 more countries, in a period from now until 2030.
In order to do so, vector control is one of the most important pillars. The use of insecticide-treated nets and indoor residual spraying, as well as drugs is crucial. This year, 3 pilot countries (Ghana, Kenya and Malawi) are introducing malaria vaccination1.
On this World Malaria Day, we want to raise awareness about the disease and its effects both in our cells and in a general perspective because, as this year’s key message reads, Zero malaria starts with me.
Bibliography:
- World Health Organization (https://www.who.int/campaigns/world-malaria-day/world-malaria-day-2019)Gomes, P. S., Bhardwaj, J., Rivera-Correa, J., Freire-De-Lima, C. G., & Morrot, A. (2016). Immune Escape Strategies of Malaria Parasites. Frontiers in microbiology, 7, 1617. doi:10.3389/fmicb.2016.01617
- Moore, L. R., Fujioka, H., Williams, P. S., Chalmers, J. J., Grimberg, B., Zimmerman, P. A., & Zborowski, M. (2006). Hemoglobin degradation in malaria-infected erythrocytes determined from live cell magnetophoresis. FASEB journal : official publication of the Federation of American Societies for Experimental Biology, 20(6), 747–749. doi:10.1096/fj.05-5122fje
- Bousema, T., & Drakeley, C. (2011). Epidemiology and infectivity of Plasmodium falciparum and Plasmodium vivax gametocytes in relation to malaria control and elimination. Clinical microbiology reviews, 24(2), 377–410. doi:10.1128/CMR.00051-10
- Shahinas, D.; Folefoc, A.; Pillai, D.R. Targeting Plasmodium falciparum Hsp90: Towards Reversing Antimalarial Resistance. Pathogens2013, 2, 33-54.
